 
The following plot shows the variation of first ionization energies across the periodic table. Consistent with size trends, first ionization energies generally increase across a period and decrease down a group. The valence electrons are closer to the nucleus to which they are attracted in a smaller atom thus, more energy will be required to remove an electron by ionization. Trends in first ionization energies can be understood on the basis of size of atoms. \Īll ionization energies are positive, because it takes energy to remove an electron from the attraction of an atom’s nucleus. The first ionization energy, \(I_1\), refers to removing one electron from a neutral atom: Ionization energy, I, is the energy required to remove an electron from a gaseous species. In the chart below, note the trends in the sizes (radii in picometers, pm) of the following ions with the same charge. Yes What are periods for periodic tables a Periodic Table organises the different elements, according to the atoms mass, type, properties, reactivity etc. Ionic radii increase down a group for ions of the same charge. Francium is named the country, _.Ģ0) _ is the second most abundant element in the universe.Ģ2) The element named after Nobel Prize winning scientists, Marie Curie and Pierre Curie.Ģ5) The noble gases all share this property.Ģ6) Atoms of a particular element are identified by their number of _.Ģ7) Mercury and Bromine are both _ at room temperature.Ģ8) The element that can form over 10 million different compounds.ģ1) Alkaline Earth metals have two of these.ġ) Element named after the greek word “Argos” meaning “idle” or “lazy”.Ģ) The element represented by the symbol W.ģ) Brass is an alloy, made of two elements, copper and _.ĥ) Diamond and graphite are both _ of carbon.ħ) The most abundant element in the universe.Ĩ) The most abundant element in the Earth’s crust.ġ2) The element name Helium is derived from a Greek word meaning _.ġ5) The element that makes up approximately 50% of bone mass.ġ7) An element that is a crucial building block of DNA.Ģ1) He organized the periodic table by atomic weight.Ģ3) The latin word for Tin, as indicated by its element symbol.Ģ6) A radioactive heavy metal, often considered as the most dangerous element.Ģ9) A popular poison in history, often known as “inheritance powder”.ģ0) A soft slivery metal, often used by magicians.ģ2) The most abundant element in the human body (by mass).\): 69 pm (Hint: it has a single-letter element symbol.)ĩ) A group of elements are also called a _.ġ2) Sterling Silver is an alloy made of two elements, silver and _.ġ4) _ was the first man-made element.ġ6) The acronym responsible for maintaining the periodic table.ġ8) Several elements are named related to places on Earth. The Periodic Table Trivia: Crossword CluesĤ) 90 of the elements on the periodic table can be found in _.Ħ) The heaviest, naturally occurring element. Periods 6 and 7 have 32 elements, because the. Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements. A new period begins when a new principal energy level begins filling with electrons. That may be of interest to teachers as they plan to teach topics related to the periodic table. There are seven periods in the periodic table, with each one beginning at the far left. 
There is a Periodic Table Unit Plan available in the AACT library.An answer key document has been provided for teacher reference.The AACT Video Dimitri Mendeleev may be helpful for reference. 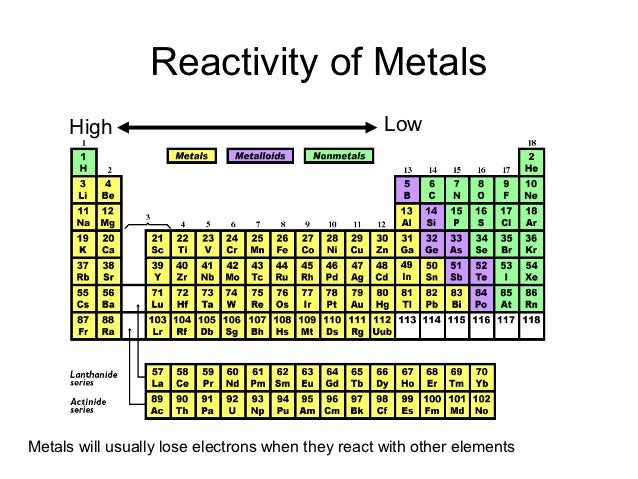
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
